LEDs + Lighting
Neon: A Light History
ST has covered many neon-lighting developments in its 100 years, but neon predates even the magazine.
Published
18 years agoon
Over its 100 years of continuous publication, ST has reported on many new signmaking technologies and their development, including neon.
Some might say ST predates neon signs, but this applies only to neon's widespread public use. In 1857, German glassblower Heinrich Geissler bent glass tubing filled with gas and illuminated the letters (used in shop windows) by applying high-voltage electricity (see ST, December 2001, page 20). However, in this centennial tribute, I won't discuss "who's first," but I'll present a short, incomplete kaleidoscope of developments in more than 100 years of luminous-tube signage.
In the second half of the 19th Century, partially evacuated glass vessels fitted with electrical connections were used only in scientific research, but this spawned the discovery of atomic structure and, eventually, quantum mechanics (the basis of our semiconductor industry).
Early 20th-Century breakthroughs in glass and electrical technologies led to the electron tube, radio and television. This ultimately laid the foundation for information technology, the third industrial revolution.
In the early 20th Century, D. McFarlan Moore's large-diameter (approximately 2-in.) tubes were rarely incorporated into advertising signs due to problematic manufacturing and installation. Also, impure gases that chemically reacted during operation shortened their operating life. Even a reservoir and automatic refill valves couldn't prevent the fill gas from being quickly consumed.
The breakthrough occurred in 1910, when Frenchman Georges Claude patented the use of neon gas in glass tubing and a minimum, electrode-surface size for a given electrical current. More businessman than inventor, Claude realized the advertising potential of an intense red light and successfully promoted bent neon tubes as signs.
Vacuum technology
During Geissler's and Moore's days, vacuum technology was quite complicated. In the 19th Century, manually operated piston pumps (Fig. 1) couldn't achieve an adequately low pressure, so Geissler improved Sprengel's mercury droplet pump, in which a row of droplets was propelled by the weight of the mercury column against the barometric pressure. The pump, operated by periodically lifting the mercury into the reservoir, produced a high vacuum, but the process required significant time. Also, mercury vapor remained in the vessel.
Claude's patent depicted the precursor of today's fastest and cleanest vacuum pumps, the cryo-sorption pump (Wolfgang Gaede hadn't invented the rotary-vane mechanical pump yet). Having generated a preliminary vacuum with a Sprengel pump, Claude connected the neon tube to be processed to a vessel filled with purified charcoal, then immersed the vessel into liquefied air. At approximately -292° F, the remaining gases condensed.
After the charcoal absorbed the impurities released by bombarding (Claude had already introduced high-voltage bombarding), the tube was sealed off the charcoal vessel. According to Claude, the time-consuming cleaning process had to be repeated until the tubes "yield the current," and the charcoal had to be prepared and cleaned every time.
In 1912, shortly after Gaede's rotary-vane pump debuted, Irving Langmuir invented the diffusion pump, which quickly obtained the low pressure required to process clean neon tubes. When the first neon-signmaking books were published in the early 1930s (after Claude's patent had expired), the diffusion pump — in its most basic form — had already established its place in every adequate, neon-processing system.
The early diffusion pump needed a trap to prevent the mercury vapor from streaming back into the sign tube. During World War II, the demand for a high-vacuum apparatus spurred the development of high-speed, multi-stage diffusion pumps, and special oils replaced mercury to operate those pumps.
AdvertisementThe new oils made the freeze-trap obsolete. In the 1960s, mineral oils were replaced by silicone-based fluids (even today, they're mostly manufactured by Dow Corning), which were much more stable against exposure to air and oxygen than mineral oils, and yielded a higher vacuum.
Gaede and Holweck invented the turbomolecular pump in the 1920s, but neon signshops didn't adopt it until the early 1980s, because early pumps couldn't withstand harsh, neon-shop conditions. Electronically controlled, high-frequency induction motors (which operated at more than 90,000 rpm), magnetic bearings and Arthur Pfeiffer's new blade materials enabled fast and reliable pumps, but they cost more than the standard diffusion pump. The turbomolecular pump delivered optimum pumping power only in very low vacuum ranges, and it didn't pump fast enough to accommodate the end of the bombarding process.
In the early 1990s, Alcatel, a French company, introduced the "turbo-drag" pump, which combined the turbomolecular pump with a principle mentioned by Gaede, and yielded high pumping speed, at approximately 1 mbar pressures, required for the end of the bombarding process. Turbo-drag pumps are today's state-of-the-art equipment for neon- and cold-cathode-processing, but many rudimentary, U.S. signshops still use only a two-stage, rotary-vane pump, which, if kept in perfect condition and skillfully operated, can yield small-volume neon tubing that operates more than 50,000 hours without trouble.
Electrode history
Long-lasting, neon-tube electrodes have always been scarce. Early 19th-Century researchers found, as an electrode material, pure aluminum didn't disintegrate as quickly as iron or copper. Early, rod-like electrodes measured approximately 1⁄8 in. in diameter and in. long. Electrostatic generators and small Rumkorff induction coils produced currents that measured only fractions of a milliampere.
Higher currents, necessary to produce large amounts of light, quickly destroy electrodes because of sputtering (an abnormal glow discharge with a high, cathode fall). Claude found, if you want to use a high current, the electrode surface must be enlarged, and a minimum surface-per-current unit must be provided to give the electrode and tube a long life.
AdvertisementClaude suggested copper or graphite as electrode material, but I've seen Claude electrodes that only used large, graphite rods. Claude safeguarded his patents by keeping the knowledge and materials for manufacturing neon signs in a closed community, becoming part of it required a $100,000 investment.
Thus, some tried to circumvent the patents. Ben Kresge and the Cortese brothers introduced a completely different electrode design in which the interior of a hollow-iron cup was coated with earth alkali oxides, using a 1907 patent of Arthur Wehnelt, a German physicist, which permitted a much higher current per surface than Claude had claimed without sputtering.
The coating's composition has been slightly changed, but today's electrodes are virtually the same. An addition introduced in the late 1940s, a ceramic collar, covers the front edge of the open metal cup to corral the sputtering process inside the cup so that the glass surrounding the metal part remains clean awhile.
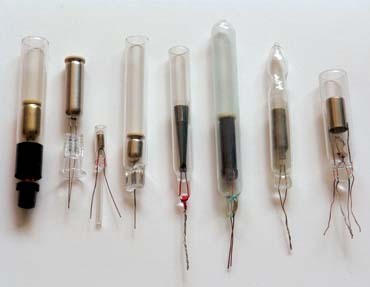
Throughout the years, different shell shapes facilitated processing and extended tube life. In 1910, Claude had suggested wrapping electrical and thermal-insulating material around the electrode body, like a sheet of mica or glass beads, to prevent it from touching the glass. We've seen such curiosities as the "shell inside a shell," the "helium-resistant coating" or the "800mA filament and shell" electrode (Fig. 2), but none gained a wide marketshare. Nevertheless, coated electrodes entail more difficult processing. Some countries still use the old, uncoated electrodes (comprising oxidized iron, not carbon).
Transformers
Neon signs evolved shortly after electrification's heyday. Debate centered on the adoption of alternating current (supported by Westing-house, Nicola Tesla and GE) or direct current (Thomas Alva Edison's pick). Neon tubes need high voltage to operate, but the current must be limited by the power source to protect the tube. The early, electrostatic machines provided direct current, but, for the widespread supply of electrical power, alternating current was the first choice.
Alternating current prevailed because it allows low-voltage current to be easily stepped up to high voltage using a transformer that contains only copper-wire windings and an iron core, while direct current would need a rotating motor-generator to step up the voltage.
Today's transformer materials, especially the plastics that insulates the wires, varnishes and epoxy-resin compounds, have improved. Formerly, sheets of mica and oil paper insulated the copper windings, and wax or tar encased the interior to avoid moisture damage. Although we now try to extract more power from a smaller transformer, some claim the "old style" transformers were sturdier and would last longer than the new ones.
In the early 1980s, power semiconductors, and thus, electronic neon power supplies, debuted on the market. Electronic power supplies operate on high-frequency, high-voltage electricity, but many end users learned the hard way that electronic power supplies fit certain applications, whereas magnetic transformers fit others.
Electronic power control and high frequency permitted previously unattainable special effects. But, for special effects, such as flow neon, electronic specialists didn't wait for semiconductors to emerge. Fig. 3 shows an electronic-flow, neon power supply, built in the 1960s, that exclusively uses electron tubes.
Glass, colors and accessories
Early neon signs featured four colors: blue (argon and mercury in clear glass), red (neon in clear glass), green (argon and mercury in green glass) and yellow (helium in green glass). All tubing was uncoated.
Tubing coated with fluorescent material first appeared in the early 1930s, parallel to the first, experimental, low-voltage, hot-cathode, fluorescent lamps. Since then, available colors have increased. Fluorescent materials have changed and, thus, increased the efficiency of the neon/cold-cathode tubes, and the neonbenders claim their suppliers don't report such changes.
"Old style" materials often contained such poisonous ingredients as arsenic or cadmium, but they were quite stable and yielded a low light output. Especially for the whites, these "classic" fluorescent materials were replaced by halophosphates, which have proven more efficient and give an acceptable color rendering, but they require careful processing and lose intensity rather quickly.
Television-screen and fluorescent-lamp research led to the "rare-earth" phosphors, which mix red, green and blue powders to produce whites up to 90 lumens per watt for cold-cathode tubing at a very good color rendering. This surpasses, by more than two times, the efficiency of a good, white LED and is surpassed only by hot-cathode lamps (fluorescents). Abundant fluorescent materials and glasses combine to produce more than 140 neon-color choices.
Other materials have changed, too. One major leap occurred in the mid-1990s, when an Italian company introduced a new glass composition that behaved similarly to traditional lead glass but didn't contain environmentally questionable ingredients.
Approximately 15 years ago, major glass manufacturers discontinued colored glasses that produced rich and saturated colors. Different sources now offer colored, soft-glass tubing, thanks to Venetian glassmakers who had first filled the demand. However, the beauty of some old colors has never been reached again.
SPONSORED VIDEO
Introducing the Sign Industry Podcast
The Sign Industry Podcast is a platform for every sign person out there — from the old-timers who bent neon and hand-lettered boats to those venturing into new technologies — we want to get their stories out for everyone to hear. Come join us and listen to stories, learn tricks or techniques, and get insights of what’s to come. We are the world’s second oldest profession. The folks who started the world’s oldest profession needed a sign.
You may like
Advertisement
Subscribe

Magazine
Get the most important news
and business ideas from Signsofthetimes Magazine.
Advertisement
Most Popular
-

 Tip Sheet4 days ago
Tip Sheet4 days agoAlways Brand Yourself and Wear Fewer Hats — Two of April’s Sign Tips
-

 Business Management2 weeks ago
Business Management2 weeks agoWhen Should Sign Companies Hire Salespeople or Fire Customers?
-

 Women in Signs2 weeks ago
Women in Signs2 weeks ago2024 Women in Signs Award Winners Excel in Diverse Roles
-

 Real Deal5 days ago
Real Deal5 days agoA Woman Sign Company Owner Confronts a Sexist Wholesaler
-

 Benchmarks1 day ago
Benchmarks1 day ago6 Sports Venue Signs Deserving a Standing Ovation
-

 Editor's Note1 week ago
Editor's Note1 week agoWhy We Still Need the Women in Signs Award
-

 Line Time2 weeks ago
Line Time2 weeks agoOne Less Thing to Do for Sign Customers
-

 Product Buying + Technology1 week ago
Product Buying + Technology1 week agoADA Signs and More Uses for Engraving Machines